Deserts are some of the most extreme habitats on the planet. The Sahara, the Gobi and the Sonora are some examples of warm deserts where the high temperatures and the lack of water pose a great challenge to animals that live in them. Reptiles are one of the animal groups that present the most incredible adaptations for life in deserts. In this entry we’ll explain the difficulties that desert reptiles must face in order to survive, and we’ll introduce you to different species of snakes and lizards that in the deserts have found their home.
REPTILES IN THE DESERT
The characteristic which unites all deserts is the scarce precipitation as, unlike most people think, not all deserts present high temperatures (there are also cold deserts, like the Arctic and the Antarctic, both in danger because of the climate change). Reptiles thrive better in warm deserts than in cold deserts, because the low temperatures would not allow them to develop their life activity.

Warm deserts not always have extremely high temperatures. While during the day temperatures may rise up to 45°C, when the sun goes down temperatures fall below freezing point, creating daily oscillations of up to 22°C. The different desert reptiles, being poikilotherms and ectotherms, use different behavioural strategies in order to avoid overheating during the day and to keep their body heat during the night (for example, climbing to elevated areas or sleeping in burrows).
The Namaqua chameleon (Chamaleo namaquensis) regulates its body heat changing its colouration. During sunrise it is black in colour in order to absorb as much radiation of the sun and activate its metabolism. When temperatures become higher, it turns white to reflect solar radiation. Video from BBC.
As we have already stated, the main characteristic of any desert is the lack of water. Generally, in a desert, it rains less than 250 mm of water a year. The scaly and impervious skin of reptiles prevents the loss of water, and their faeces contain uric acid which, compared to urea, is much less soluble in water, allowing them to retain more liquids. Most desert reptiles extract the water they need from their food and some drink water from the dew.
Both the extreme temperatures and the shortage of precipitations make the desert a place with very few living beings. Vegetation is scarce and animals are usually small and secretive. This lack of resources causes desert reptiles to be usually smaller than their cousins from more benevolent environments. Also, these saurians usually exploit any available food resource, although they think twice before wasting their precious energy to get their next meal.
SAND SNAKES
In many sandy deserts we can find various species of snakes (and legless lizards) that have adapted to a life among the dunes. Many of these ophidians share a locomotion method called “sidewinding”, in which they raise their head and neck from the ground and move them laterally while the rest of the body stays on the ground. When they place their head on the ground again they raise their body, making these snakes move laterally in a 45° angle. This method of locomotion makes these snakes move more efficiently in an unstable terrain. It also reduces the contact of their body with an extremely hot substrate, as the body of these ophidians only touches the ground in two points at a time.
As we can see in this video from RoyalPanthera, sidewinding allows desert snakes to move minimizing the contact with the hot terrain.
Many desert ophidians bury themselves in the sand both to avoid sun exposure and to blend in and catch their prey unaware. This has made many desert-dwelling snakes very sensitive to vibrations generated by their prey as it moves through the sand. In addition some species present an overly developed rostral scale (the scale at the tip of their snout), being much thicker in order to aid during excavation in sandy soils.

The horned vipers of the Cerastes genus also present various characteristics that facilitate life in the deserts. These vipers evade high temperatures becoming active at night and they spend the day buried in the sand. Their hunting method consists in burying themselves waiting for a prey to pass by, this way saving most of their energy. It is believed that their horn-shaped supraocular scales cover their eyes when they are buried in order to protect them from the sand.

SPINY CRITTERS
In different deserts of the world we find reptiles with their bodies covered in spines. This not only provides them with certain protection against predators, but is also helps them blend in in a habitat with plenty of thorny plants. Two of these animals are members of the Iguania suborder: the thorny devil and the horned lizards.

The thorny devil (Moloch horridus) is an agamid that lives in the Australian sandy deserts. This lizard presents spines all over its body, making it difficult for its predators to swallow. It also has a protuberance behind its head that acts as a fat storage. When it feels threatened, it hides its real head between its legs and it exposes its neck protuberance as a decoy head. Probably, the most interesting adaptation of this animal is the system of small grooves among its scales, which collect any water that contacts its skin and conducts it directly to its mouth.
Horned lizards (Phrynosoma genus, affectionately called “horny toads”) are iguanids which are found in different arid habitats of North America. Similarly to the thorny devil, their body is covered in spines making them hard to eat for their predators. Also, when they are caught, they inflate their bodies to make the task even more difficult. Finally, some species like the Texas horned lizard (Phrynosoma cornutum) are known for their autohaemorrhagic abilities: when they feel cornered they squirt a stream of stinky blood from their eyes which scares away most predators.

As you have seen, in the deserts we can find reptiles with some of the most inventive (and disturbing) adaptations of the world. These are only a few examples of the astonishing diversity of squamates that are found in the deserts of the world, which only seek to survive the harsh conditions of these extreme environments. Sometimes, it’s just a matter to avoid burning your feet with the hot sand.
Video from BBCWorldwide of a shovel snouted lizard (Zeros anchietae) making the “thermal dance” in order to diminish the contact with the hot sand.
REFERENCES
The following sources have been consulted during the elaboration of this entry:
- Halliday & Adler (2007). La gran enciclopedia de los Anfibios y Reptiles. Editorial Libsa.
- Digital-Desert. Desert Reptiles.
- Arizona-Sonora Desert Museum. Adaptations of Desert Amphibians & Reptiles.
- In the desert. A comprehensive list of the venomous snakes found in the desert.
- H.E.R.P. Herpetological Education & Research Project.
- Christopher J. Bell, Jim I. Mead & Sandra L. Swift (2009). Cranial osteology of Moloch horridus (Reptilia: Squamata: Agamidae). Records of the Western Australian Museum. Vol 25. Pp: 201-237.
- Horned Lizard Conservation Society.
- Cover image by Yathin S. Krishnappa.




 The venom of many animals is used for both antivenom development and pharmacological research of analgesics and other medicines. Photo of the extraction of venom from a saw-scaled viper (Echis carinatus), by
The venom of many animals is used for both antivenom development and pharmacological research of analgesics and other medicines. Photo of the extraction of venom from a saw-scaled viper (Echis carinatus), by  Indian wolf snake (Lycodon aulicus), example of an ophidian. Photo by
Indian wolf snake (Lycodon aulicus), example of an ophidian. Photo by  Brown basilisk (Basiliscus vittatus), example of an iguanian. Photo by
Brown basilisk (Basiliscus vittatus), example of an iguanian. Photo by  Earless monitor lizard (Lanthanotus borneensis), example of an anguimorph. Photo by
Earless monitor lizard (Lanthanotus borneensis), example of an anguimorph. Photo by 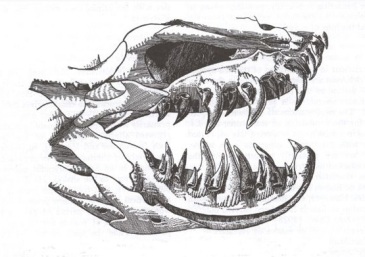 Helodermatid skull, in which we can see the sharp teeth with which they inject their venom. Image from
Helodermatid skull, in which we can see the sharp teeth with which they inject their venom. Image from  Photo by
Photo by  Photo of an eastern bearded dragon in which we can see its yellow coloured mouth. Could it be that this coloration is indicating anything? Photo by
Photo of an eastern bearded dragon in which we can see its yellow coloured mouth. Could it be that this coloration is indicating anything? Photo by 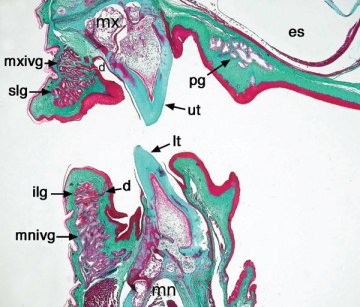 Transversal section of the mouth of an eastern bearded dragon, in which we can see the incipient venomous glands both in its upper jaw (mxivg) and its lower jaw (mnivg). Image extracted from
Transversal section of the mouth of an eastern bearded dragon, in which we can see the incipient venomous glands both in its upper jaw (mxivg) and its lower jaw (mnivg). Image extracted from  Perente or perentie (Varanus giganteus) a typical varanid, with long neck, strong legs, active metabolism and developed senses. Photo by
Perente or perentie (Varanus giganteus) a typical varanid, with long neck, strong legs, active metabolism and developed senses. Photo by  Photo in which we see the feared monitor’s saliva, specifically from an Asian water monitor (Varanus salvator). Image by
Photo in which we see the feared monitor’s saliva, specifically from an Asian water monitor (Varanus salvator). Image by  Skull of megalania (Varanus priscus) in which we can see the teeth without gooves. This extinct monitor with more than 5 metres long, was the largest venomous animal known. Photo by
Skull of megalania (Varanus priscus) in which we can see the teeth without gooves. This extinct monitor with more than 5 metres long, was the largest venomous animal known. Photo by  A group of Komodo dragons (Varanus komodoensis) feeding on a recently killed pig. Image extracted from
A group of Komodo dragons (Varanus komodoensis) feeding on a recently killed pig. Image extracted from 

 Diagram of a crocodile egg: 1. eggshell 2. yolk sac 3. yolk (nutrients) 4. vessels 5. amnion 6. chorion 7. air 8. alantois 9. albumin (white of the egg) 10. amniotic sac 11. embryo 12. amniotic fluid. Image by
Diagram of a crocodile egg: 1. eggshell 2. yolk sac 3. yolk (nutrients) 4. vessels 5. amnion 6. chorion 7. air 8. alantois 9. albumin (white of the egg) 10. amniotic sac 11. embryo 12. amniotic fluid. Image by  Diagram of an amphibian egg: 1. jelly capsule 2. vitelline membrane 3. perivitelline fluid 4. yolk 5. embryo. Image by
Diagram of an amphibian egg: 1. jelly capsule 2. vitelline membrane 3. perivitelline fluid 4. yolk 5. embryo. Image by  Reconstruction of Solenodonsaurus janenschi, one of the candidates in being the first amniote, which lived around 320-305 million years ago in what is now the Czech Republic. Reconstruction by
Reconstruction of Solenodonsaurus janenschi, one of the candidates in being the first amniote, which lived around 320-305 million years ago in what is now the Czech Republic. Reconstruction by  Diagram of an anapsid skull, by
Diagram of an anapsid skull, by  Diagram of a synapsid skull, by
Diagram of a synapsid skull, by  Diagram of a diapsid skull, by
Diagram of a diapsid skull, by  Drawing of the skull of Archaeothyris, which is thougth to be one of the first synapsids that lived around 306 million years ago in Nova Scotia. Drawing by
Drawing of the skull of Archaeothyris, which is thougth to be one of the first synapsids that lived around 306 million years ago in Nova Scotia. Drawing by  Reconstruction of Dimetrodon grandis, one of the better known synapsids, from about 280 million years ago. Reconstruction by
Reconstruction of Dimetrodon grandis, one of the better known synapsids, from about 280 million years ago. Reconstruction by  Evolutionary tree of current vertebrates, in which green color marks the groups previously included inside reptiles. As you can see, the traditional conception of "reptile" includes the ancestors of mammals and excludes birds. Image by
Evolutionary tree of current vertebrates, in which green color marks the groups previously included inside reptiles. As you can see, the traditional conception of "reptile" includes the ancestors of mammals and excludes birds. Image by  Shed skin of a rat snake. Photo by
Shed skin of a rat snake. Photo by  Photo of a tuatara (Sphenodon punctatus), by
Photo of a tuatara (Sphenodon punctatus), by  Photos of some squamates, from left to right and from top to bottom: Green iguana (Iguana iguana, by
Photos of some squamates, from left to right and from top to bottom: Green iguana (Iguana iguana, by  Drawing of the skull of the dinosaur Massospondylus in which we can see the different characteristic openings of diapsid archosaurs. Image by
Drawing of the skull of the dinosaur Massospondylus in which we can see the different characteristic openings of diapsid archosaurs. Image by  Photo of two species of modern arcosaurs: a Nile crocodile (Crocodylus niloticus) and a yellow-billed stork (Mycteria ibis). Photo by
Photo of two species of modern arcosaurs: a Nile crocodile (Crocodylus niloticus) and a yellow-billed stork (Mycteria ibis). Photo by  Skeleton of the extinct tortoise Meiolania platyceps which lived in New Caledonia until 3000 years ago. In this photo it can be seen the compact cranium without openings. Photo by
Skeleton of the extinct tortoise Meiolania platyceps which lived in New Caledonia until 3000 years ago. In this photo it can be seen the compact cranium without openings. Photo by  Individual leopard tortoise (Stigmochelys pardalis) from Tanzania. Photo by
Individual leopard tortoise (Stigmochelys pardalis) from Tanzania. Photo by 


 Image by
Image by  Foto of a Cairo spiny mouse (Acomys cahirinus), a mammal which is able to shed and regrow its tail. Photo by
Foto of a Cairo spiny mouse (Acomys cahirinus), a mammal which is able to shed and regrow its tail. Photo by  Tridimensional model of the fracture planes on the tail of a lizard and the regeneration post-autotomy of a cartilaginous tube. Image extracted from
Tridimensional model of the fracture planes on the tail of a lizard and the regeneration post-autotomy of a cartilaginous tube. Image extracted from  Photo of a Catalonian wall lizard (Podarcis liolepis) that has shed its tail. Photo by
Photo of a Catalonian wall lizard (Podarcis liolepis) that has shed its tail. Photo by  Detail of the tail of a common wall gecko (Tarentola mauritanica) which has regenerated the tail without losing its original tail. Photo by
Detail of the tail of a common wall gecko (Tarentola mauritanica) which has regenerated the tail without losing its original tail. Photo by  Wood frog tadpole (Rana sylvatica) which, like all amphibians, delays the development of its legs up to the moment of metamorphosis. Photo by
Wood frog tadpole (Rana sylvatica) which, like all amphibians, delays the development of its legs up to the moment of metamorphosis. Photo by  Diagram about the formation of the blastema in a zebrafish (Danio rerio) another model organism. Image from
Diagram about the formation of the blastema in a zebrafish (Danio rerio) another model organism. Image from  Photo of an axolotl, by
Photo of an axolotl, by 








 A male slow worm on its habitat, on the Netherlands, by
A male slow worm on its habitat, on the Netherlands, by 












